At Surescreen, the main thing we are trying to do is develop new tests for diseases beyond COVID-19, improve the sensitivity of tests so we are able to detect diseases better or earlier, and essentially increase the range of things we are able to accomplish using LFDs. My role as a Research and Development Scientist is to produce the tests themselves – all the way from doing the background chemistry and physics, through to spraying chemicals onto the strips of paper that are then packaged up within the plastic testing cassettes.
The first stage of research and development is disease targeting, which means choosing a disease with a suitable medical and business case for detection. At this point, we would be consulting and collaborating with colleagues from other medical fields – the NHS, academics, consultants and so on – to identify diseases that would benefit most from a rapid test. Those would potentially be diseases for which you need to get results quickly so that you can come up with a good treatment plan straight away, but where current testing takes a long time. Alternatively, we might be looking at diseases for which you might face regular screening once you hit a certain age category that puts you at greater risk. Any disease that creates a high input of tests and large workload for testing labs is a potential area of interest that could benefit from people taking rapid tests at home.
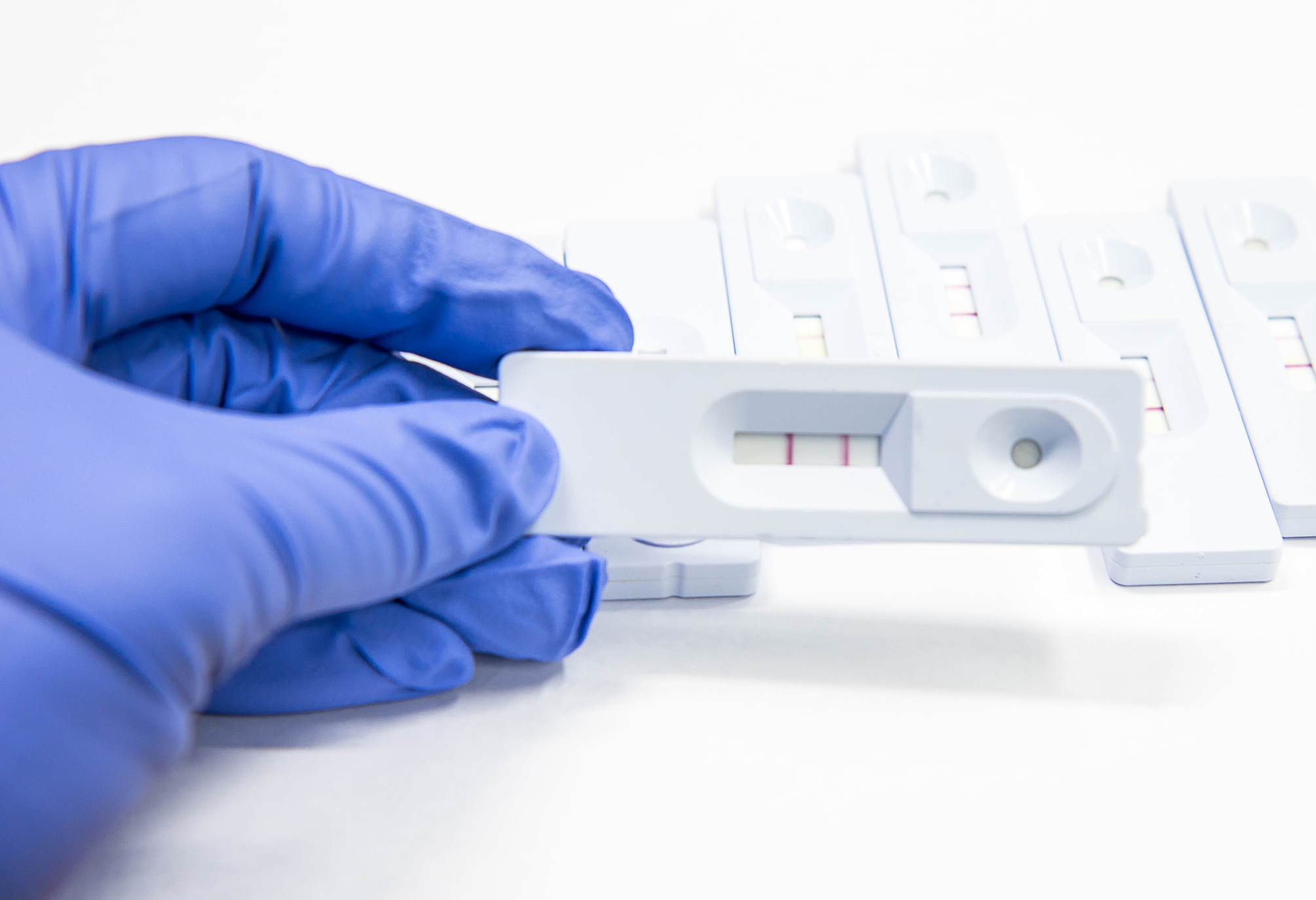
Once we’ve identified a target disease, the next stage is to obtain the antibodies that we use to develop the LFDs. In the most basic sense, an antibody is effectively a small grabber that your body produces as part of an immune response, and it will specifically bind onto another molecule – in this case a virus, bacteria, protein or similar. We spray these antibodies onto the ‘test’ line of the LFD, so that as the sample liquid flows past they will ‘grab’ onto the virus (if present). At the same time, there are also some very small bits of gold – the nanoparticles that I make – which have a corresponding antibody on them that ‘grabs’ onto the virus. The gold is coloured bright red, so if the virus is present, the gold antibody then sticks to the ‘test’ line along with the virus – giving the bright red line that indicates you have tested positive. If the virus isn’t present, the gold flows past so that there is no colour sticking to the test line – a negative result.
One of the biggest challenges during the R&D stage is achieving the correct level of specificity. Success depends on your antibody being able to pick out one – and only one – marker. Taking the COVID test as an example, you need to be extremely sure it’s only detecting COVID, and not also colds and flu. This means that whenever we pick a new target, we need reliable antibodies in order to make sure that the test we make is incredibly specific to the one marker that we’re looking for. Once we have achieved the desired level of specificity, it is then absolutely vital to test and validate our product before sending anything out, to ensure that we are achieving our goal of providing an accurate diagnosis.